PURPOSE:
The purpose of this lab is to determine the conditions in which the enzymes in a chicken liver can properly function, by testing the reaction with the substrate H2O2 and liver enzyme at different temperatures. Enzymes are known to function best in a specific temperature and pH range. The independent variable of the experiment is the heat level,, while the dependent variable is the rate of reaction.
This lab relates to the Biochemistry unit, as it demonstrates the effective range of enzymes. These enzymes bind with substrates to from a substrate-enzyme complex and catalyze reactions in the body.
This lab relates to the Biochemistry unit, as it demonstrates the effective range of enzymes. These enzymes bind with substrates to from a substrate-enzyme complex and catalyze reactions in the body.
Hypothesis:
If vial is cooled down, the liver will bubble slowly, while if it is heated, the liver will bubble even more slowly.The vial at room temperature should react the most.
MEthods Summary:
IV: The IV is the heat of the vial.
Levels of IV: The levels of Room Temperature, Warm, and Cold
DV: The DV is the rate of reaction.
Measurement: The DV is measured by observing the amount of bubbles emerging from the liver. It's a qualitive measurement with a scale of 0 for no reaction to 5 for highest rate of reaction.
Control: The controls is the vial with no liver added.
Constant: the constants are the amount of water and amount of H2O2 used.
Number of trials: There is one trial for this experiment.
DV: The DV is the rate of reaction.
Measurement: The DV is measured by observing the amount of bubbles emerging from the liver. It's a qualitive measurement with a scale of 0 for no reaction to 5 for highest rate of reaction.
Control: The controls is the vial with no liver added.
Constant: the constants are the amount of water and amount of H2O2 used.
Number of trials: There is one trial for this experiment.
Materials:
- 4 vials
- 3 pieces of liver
- H2O2
- A hot plate
- An ice bath
- A beaker
- Water
- Tongs
Procedure:
- Place a set amount of water in all 4 vials.
- Place a piece of liver in 3 of the 4 vials.
- Put one vial with liver inside an ice bath and let it cool for 15 minutes
- Fill up a beaker with water, and then place one of the vials with liver inside the beaker.
- Plug in the hot plate, and then place the beaker on the hot plate.
- Let the water heat up until it begins to boil
- While the experimental vials are heating and cooling, place a set amount of H2O2 in the remaining two vials.
- Observe and record the reactions of the vial without liver and the vial of liver at room temperature.
- Once the vial in the ice bath and the vial in the boiling water are finished cooling down and heating up respectively, take them out carefully with tongs.
- Drop a certain amount of H2O2 into these two vials, and then observe and record their reactions.
- Compare the rate of reaction of each vial.
DATA TABLE
The Data was qualitatively evaluated visually using a scale of 1, for the lowest reaction, to 5, for the highest reaction. Vials 2-4 contained liver, while vial 1 only contained water. All vials had H2O2 added to them.
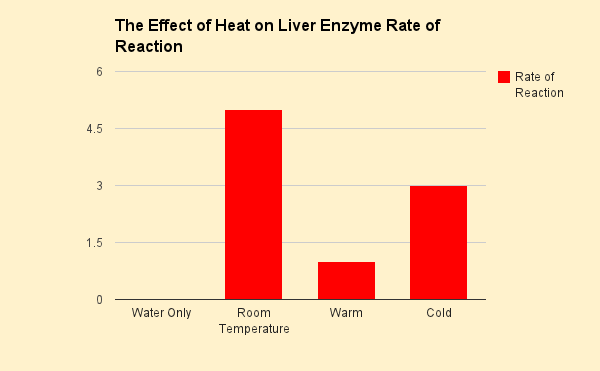
The effect of Heat on Liver Enzyme Rate of Reaction
DATA chart
conclusion
The data indicated that the vial of liver at room temperature had the highest rate of reaction, while the the cold water had a lower rate of reaction, and the heated one had the lowest rate of reaction. On the qualititative score set, room temperature had a score of 5, warm had a score of 1, and cold had a score of 3. The plain water had no reaction at all, since no liver was present in that vial. This supports the hypothesis that the rooom temperature vial would have the highest rate of reaction, while the heated one would have the lowest, and the cold vial would be in between the others with a low rate of reaction. These results likely occured because enzymes work within a certain range of temperature and pH. By leaving the enzyme at its optimal temperature, it will function with a higher rate of reaction compared to at other temperatures. Cooling down reactants generally causes the rate of reaction to slow down, so the cooled down vial had a low rate of reaction compared to the room temperature vial. Heating reactants can speed up the rate of reaction, however, too much heat will cause enzymes to denature. In this case, evidence suggests that the enzymes did denature in the boiling water, so the hot vial had an extremely low rate of reaction compared to the others. Some errors in the experimental process include not not boiling and cooling the liver for the same amount of time, not measuring the rate of reaction for the same amount of time, and not slicing the liver into equal pieces. This could have affected the rate of reaction in some of the data. In order to prevent these problems in the future, a stopwatch should be used for both heating and cooling the liver as well as measuring the rate of reaction. Extra care and some measurements should be taken to ensure that the liver are of equal size for the experiment.